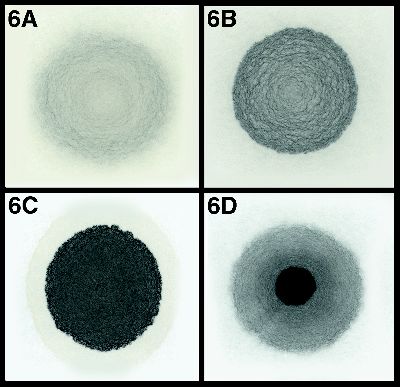
Originally Posted By: Shannow
Originally Posted By: Ducked
Not heard that one before, though there was some discussion/argument about the implications of the behaviour of oil droplets on water a while back.
IIRC with modern oils the implications for that were fairly unclear. How does ATF compare?
Drain pan is one of these...
So had a couple of square feet of black stuff, as I turkey basted the power steering fluid out of the reservoir.
The oils always "smear" into each other, "clinging" due to their surface tension as soon as they touch.
Today the Power Steering Fluid "beaded" when squirted into the collection area on top of the engine oil.
While since I read up on it, but I THINK the story with oil/water was that, as oil degrades, polar compounds develop which reduce the surface tension because they're attracted to the polar water molecules, so the oil no longer "beads" on water but spreads out as a thin surface layer. This behaviour is/was used as an indicator of oil degredation.
I suppose the ATF story might be the reverse of that. The contaminated oil is polar (like the water) and the uncontaminated ATF beads on it because it isn't.
In practice, modern oils have a lot of polar surfactants (dispersants, detergents, maybe friction modifiers) in them straight out of the bottle, so maybe the method no longer works as a reliable indicator of degredation.
www.nttworldwide.com/docs/camtel
The Role of Interfacial Tension Measurement in the Oil Industry. Carole Moules, Camtel Ltd.
Has a comparison of surface tension measurements for clean and dirty motor oils which show no systematic differences and are all very much less than those for sunflower oil.